Anhydrite
Anhydrite, or anhydrous calcium sulfate, is a mineral with the chemical formula CaSO4. It is in the orthorhombic crystal system, with three directions of perfect cleavage parallel to the three planes of symmetry. It is not isomorphous with the orthorhombic barium (baryte) and strontium (celestine) sulfates, as might be expected from the chemical formulas. Distinctly developed crystals are somewhat rare, the mineral usually presenting the form of cleavage masses. The Mohs hardness is 3.5, and the specific gravity is 2.9. The color is white, sometimes greyish, bluish, or purple. On the best developed of the three cleavages, the lustre is pearly; on other surfaces it is glassy. When exposed to water, anhydrite readily transforms to the more commonly occurring gypsum, (CaSO4·2H2O) by the absorption of water. This transformation is reversible, with gypsum or calcium sulfate hemihydrate forming anhydrite by heating to around 200 °C (400 °F) under normal atmospheric conditions.[5] Anhydrite is commonly associated with calcite, halite, and sulfides such as galena, chalcopyrite, molybdenite, and pyrite in vein deposits.
Anhydrite is one of the minerals that make up the rocks. Chemical structure similar to Gypsum but does not contain water. Formula is CaSO4.There is a sedimentary basin in large areas where sea water evaporates. It is usually found in salt deposits with gypsum next to it. Anhydrite may form on the coastline or tidal flat deposits caused by the evaporation of seawater.
Occurrence of Anhydrite
Calcium sulphate from an aqueous solution is deposited as crystals of gypsum, but when the solution contains an excess of sodium or potassium chloride, the anhydride is deposited when the temperature is above 40 ° C (104 ° F). This is one of the various methods in which the mineral is artificially prepared and is the same as its origin in nature. It is common in mineral salt basins.The anhydride is also formed as a vein filling mineral in hydrothermal deposits. The solution is usually left as a gangue in sulphide mineral deposits with calcite and halite. The anhydrite is also present in the cap rock and trap rock cavities of the salt domes.
Uses of Anhydrite
Sulfuric acid production
The paper uses anhydride as a filler. Good examples of this mineral are rare and desirable for collectors.
Tillage. Substrate and other construction materials
Anhydride, some uses are the same as Gypsum. Both minerals are crushed for use as a soil treatment and for this purpose the anhydrite is superior.
Gives more calcium per tonne in soil application. The anhydride also has a higher solubility which helps it quickly benefit the soil.
Anhydrite is used as a drying agent in paints and gypsum.
It is used in construction industry such as plaster and wall.

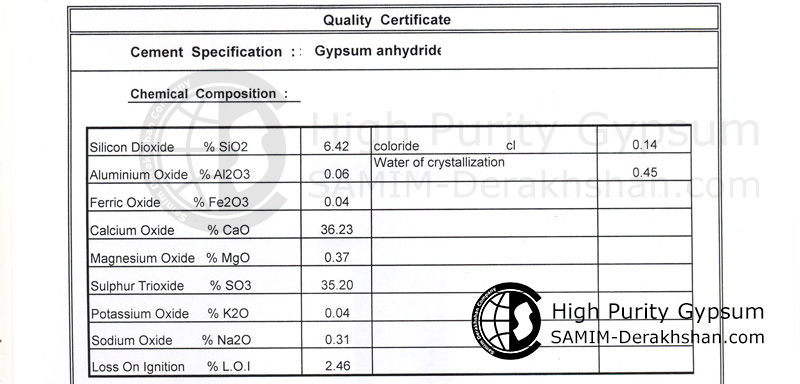
Chemical Properties of Anhydrite
| Chemical Classification | Sulfate mineral |
| Chemical Composition | CaSO4 |
Physical Properties of Anhydrite
| Color | Colorless to pale blue or violet if transparent; white, mauve, rose, pale brown or gray from included impurities |
| Streak | White |
| Luster | Pearly |
| Diaphaneity | Transparent to translucent |
| Mohs Hardness | 3.5 |
| Specific Gravity | 2.97 |
| Diagnostic Properties | Some specimens fluoresce; many more fluoresce after heating |
| Crystal System | Orthorhombic |
Optical Properties of Anhydrite
| Crystal Habit | Rare tabular and prismatic crystals. Usually occurs as fibrous, parallel veins that break off into cleavage fragments. Also occurs as grainy, massive, or nodular masses |
| Cleavage | perfect |
| Color / Pleochroism | violet varieties |
| Twinning | Simple |
| Optic Sign | Biaxial (+) |
| Birefringence | 0.042–0.044 |